Car horn installation instructions
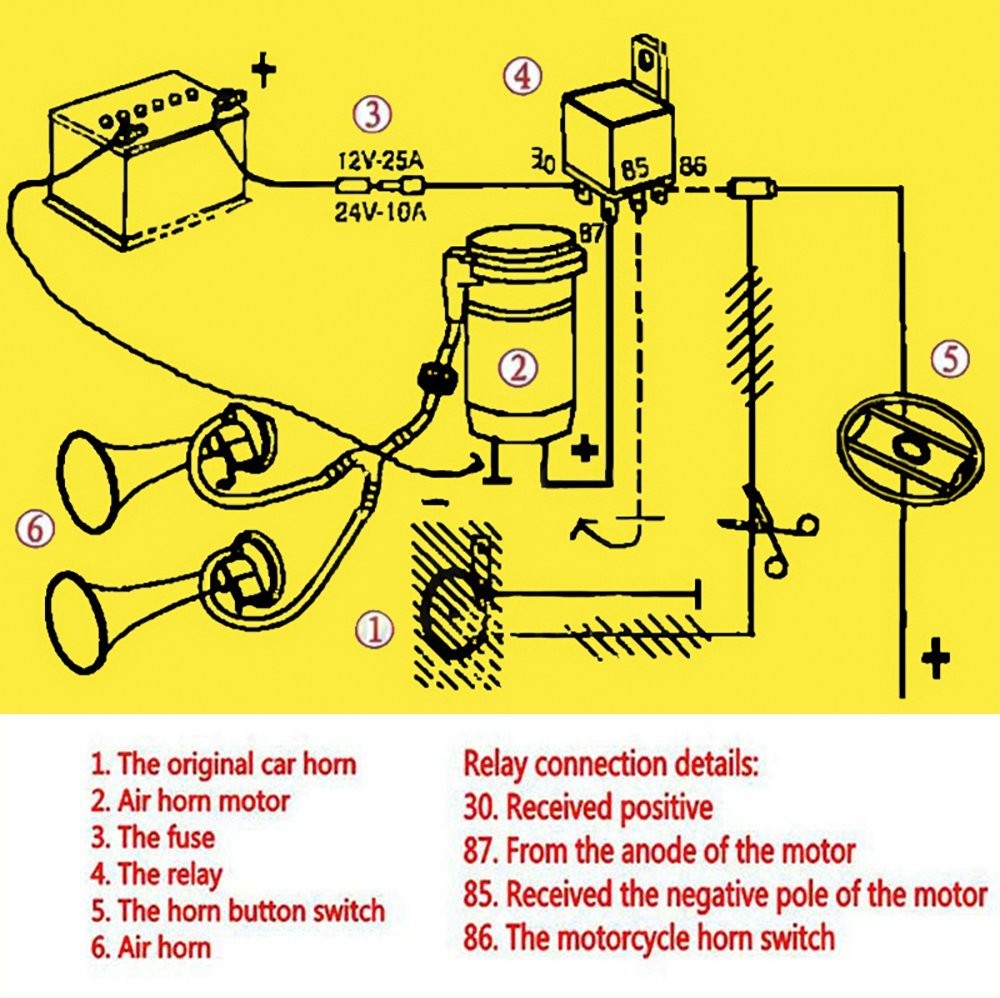
INSTALLING AFTERMARKET HORNS – ET-208. Check this diagram first to remind yourself of the original horn wiring. You will notice that power is supplied to the horn from the fuse which is always hot (not switched), so the horn will work even when the key is switched off.
Dec 05, 2018 · How to Install an Air Horn Step 1. Open the hood of your vehicle and choose a good place to mount the horns. Step 2. Using self-tapping screws and the provided mounting hardware,… Step 3. Mount the compressor to a fender or firewall with the appropriate hardware,… Step 4. Use a …
Components and Installation Instructions General Information: Your vehicle must have a 12 volt power source to hook up this horn unit. (NOTE: Our horns are NOT waterproof, so be sure to mount it in a dry location) You can wrap your horn in a plastic bag after it is …
Train Horns, Air Horns, Onboard Air Systems, and Air Suspension, we have it all! We bring the real sound of a locomotive right to your vehicle and service all your pneumatic needs. Our Horn Kits are simply ridiculous and provide your vehicle with a voice on the road!
Feb 21, 2015 · basic wiring guys, hope you guys like whats going on, we will make progress together!! i will show a more advanced circuit with relays in the future.
May 30, 2013 · How To Wire Air Horns / Air Horn Install for automotive and marine use *Includes Install instructions. Air Horn Car Install and Relay Wiring Instructions – Duration:
Dorman Products – 85929 : Electrical Switches – Specialty – Horn Button Flush Mount -. We offer a comprehensive line of Horn Buttons for a wide range of applications. All our Horn Buttons are constructed of high-quality materials for long-lasting durability.
How to Install the Air Horn on a Silverado Truck by Mike Frees . If you are not satisfied with the sound of the horn in your Chevy Silverado, replacing it with an air horn is not a difficult task. A variety of horns are available; some are one piece, with the horn and compressor assembled together. Wolo Manufacturing: Installation
Oct 09, 2019 · How to Install a Car Alarm. Car alarms are an effective way to insure your investment and give you peace of mind. While many cars come standard with some kind of alarm system, some do not. It is no surprise that many people want to install…
Oct 18, 2018 · Buy Wolo (430) Musical Horn – 12 Volt, Plays Dixieland: Air Horns – Amazon.com FREE Kindly refer the “Installation Instructions” and the “Product Description” given below for troubleshooting steps. minus the horns, and run the compressor, once with the car off, and once with the car fully on. If it works with the car off, it doesn’t
Manuals & Schematics HornBlasters
https://www.youtube.com/embed/8ZBlo52ffp0

How to wire a car horn YouTube
Welcome to HELLA Horns. Please choose your region and language! Europe deutsch english français italiano español nederlands türkçe. Asia english 中国的
Jun 26, 2017 · Air horn setups usually contain multiple horn pieces that emit sound. Because these pieces are trumpet-shaped, we’ll refer to them simply as “trumpets”. For example, kits we offer contain between 1 and 5 trumpets. Most of them also include a small air compressor. This detail will be noted on the product page.
HornBlasters Instruction Manuals and Schematics. Lose your manual or want a digital copy? We got your back! Most manuals are provided in Adobe PDF format only.
Replace your old horn in your car, boat, RV or other vehicle with this set of 12 volt electric horns, fashioned in the classic snail shape. Constructed of high-impact thermoplastic on the inside and chrome on the outside, the electric horns generate a blast that exceeds 110 decibels. Mounting tabs, brackets and installation instructions included.
Viking is a premium line of automotive horns. We carry high quality made products. Truck horns, musical horns, loud train horn kits, car horns, air horns, marine horns, sirens, and more…
Mar 08, 2016 · You will need to show us the photo of the stock horn wiring or a complete wiring diagram of the car to advise you whether the stock horn comes with relay. You want to buy a pen, you must also let us know the specs. Like type of ink, colour required, rubber grip, cap type or click type.
Mar 18, 2018 · Buy GAMPRO 12V 150db Air Horn, Chrome Zinc Dual Trumpet Air Horn with Compressor for Any 12V Vehicles Trucks Lorrys Trains Boats Cars Vans: Air Horns – Amazon.com FREE DELIVERY possible on eligible purchases

Step 1. Purchase your universal horn button kit. Note whether you want a double wire horn button or a single wire button. The single wire button will require only one wire connection, which will be a hot (positive) wire to the fuse block or to original horn wire in the horn wiring loom.
Find a good spot under the hood to install the horn(s). The ideal mounting position is towards the front of your vehicle, and pointing a bit downward so that water doesn’t collect in the horn. Mount the horn to your vehicle, using the supplied mounting hardware and screws. If necessary, install the included compressor to your firewall or a fender. Hook the compressor up to the horn with the included tubing, making sure …
Mar 08, 2015 · No need to disconnect the battery. If your car horn max at 11A, while your horn fuse is at 10A, you can just swop out the fuse for 15A one. I did that for my Stebel Magnum horns, been using them for the past 6 years without problem. If you are worried the wires may burn, then you can get the workshop to install the relay.
Hella® – Horns Installation Instructions Warranty Hella, Inc.’s limited warranty guarantees Hella products to be free from defects in materials and workmanship for the period of (1) one year after retail purchase from an authorized Hella dealer.
Our horns can also be hooked up to a 12 volt lantern battery for portability. Remote control comes complete with installed battery and operates up to a range of 10-20 feet. Quick and easy installation…takes about 5 minutes to install…instructions are included. This is the gift of a lifetime!
HWK-1 comes with wire harness, quick relay connect plug, terminals, in-line fuse, plastic wire ties and easy to follow instructions in English and Spanish. Wolo’s new wiring kit makes installation of any low pressure air horn easy
Follow these instructions (form 97524-00-01) for its use and your problem will be solved. How can I keep the horn from blowing during installation of my Grant wheel and kit? Be sure to remove the horn fuse or disconnect your battery before installation. Remember to reinstall fuse or reconnect battery when done.
Car horn installation using the example of an Audi TT/RS: Instructions Preparatory work The power supply of the acoustic signaling device is to be cut off throughout the entire setting up of the electrical connection.
We include easy-to-understand instructions and all mounting hardware and brass fittings necessary for installation. The loud 149-decibels 4-trumpet air horn kit is versatile, powerful and built to last. This horn is a perfect replacement or auxiliary horn for added safety and style.
Installing a horn button on a steering wheel may seem like a time intensive task, but in actuality, it is not that difficult. The folks at Ididit, Inc. put together a video showing the installation of a horn button on a 1955-68 GM steering wheel and horn button onto one of their Ididit, Inc. steering columns.
Dec 18, 2017 · Air Horn Car Install and Relay Wiring Instructions – Duration: 6:26. Strange Garage 83,143 views
Godzilla Roar Sounds Wireless Car Horn. These horns have a small design…but packs a big powerful sound. 20 watts output sound — 119 decibels…these horns are LOUD! Quick and easy installation…takes about 5 minutes to install…instructions are included.
Hello! I have an extra car horn and I need to install it into my car alongside my other horn, and I have no idea how to wire it! Here is what I have: a Car horn with two wires (black and red, and red) 16G wire, a switch, and a inline fuse.
Mar 30, 2016 · How to Fix a Car Horn by Mia Bevacqua on March 30, 2016 Schedule Horn does not blow Inspection. Install the new fuse – If you find the fuse has failed, install a new one of the same amperage rating. To do this, simply push the fuse back into its correct slot. Test the ground side of the circuit – According to your car’s manual
Car horns and trumpet horns The HELLA Group specializes in innovative lighting systems and vehicle electronics. It has been one of the technology leaders over the last hundred years and an important partner of the automotive industry, the independent aftermarket and for workshops.
GMTRK-2 BOLT-ON TRAIN HORN SYSTEM INSTALLATION INSTRUCTIONS For 2007-2013 GM & GMC Trucks & SUVS The GMTRK-2 System comes pre-assembled with air compressor, air tank and all fittings installed in the tank and on the battery tray plate. The horns will be bolted to the tow hook mount, making installation fast & efficient.
Using the self-tapping screw, mount the 20-amp circuit breaker in a protected location near the vehicle battery. Step 3 Connect one terminal on the circuit breaker to the positive terminal of the battery, using a piece of wire and two solderless ring terminals. Step 4
How to Install a Universal Horn Button It Still Runs
Aug 10, 2016 · In this video, the little broski and I install some new horns on the Golf. The stock horns on the car worked, however they weren’t loud enough for our liking, so we decided to pick up a set of
The Stebel Nautilus is one of my personal favorite car horns and I have owned several over the years. It has a unique sound from the twin tone harmonized horn that outputs at 139dB.It is a compressor based car horn (water resistant) that has a compact design to fit in all locations under the bonnet.
Instructions for Club Car DS Models Figure 2 Figure 1 (28905)! 2 Use part number 6439 turn signal head, that will have a corresponding nine pin connector and horn button for a plug and play installation. Use the supplied brake lead harness for switch connection. On some cars you may
How to Install the Audiovox Prestige Remote Start by Breann Kanobi . Remove starters typically install in automatic vehicles only and cannot be installed in a vehicle with a manual transmission. Step 1. How to Wire a Car Horn. How to Program a CTS Keyless Entry
Install the horn relay (included with horn kit) near the horn compressor with the electrical connectors pointing downward. Hold the relay in the desired location and use a pencil to mark the relay’s mounting holes. Set the relay aside and use an electric drill with a 1/8 inch drill bit to drill holes in the marked area.
Our 1A Auto Video Library has thousands of how-to auto repair installation videos specific to year, make and model. Each video guides you with step-by-step instructions to empower you to tackle your own repairs on your car, truck, SUV, or van.
Components and Installation Instructions
How to Install Aftermarket Hella Horns YouTube
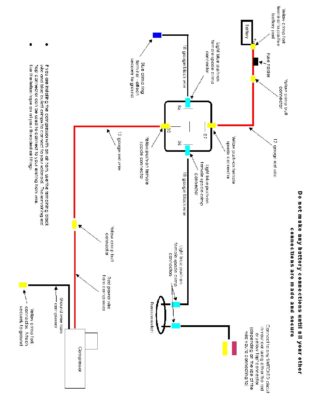
Amazon.com Wolo (430) Musical Horn 12 Volt Plays
https://www.youtube.com/embed/TYA4e2i1Jfk
Car horns and trumpet horns ─ Broad product range HELLA

Installing aftermarket horns
How to Wire and Install Hella Horns 1A Auto
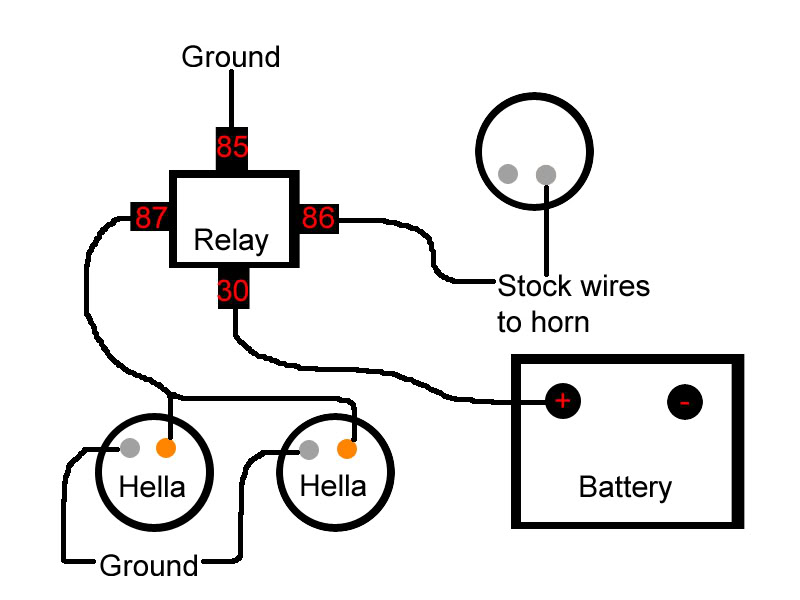

2 Piece 12 Volt Electric Horn Set Harbor Freight Tools
https://en.wikipedia.org/wiki/Vehicle_horn
How To Install A Car Horn Button Speedway Motors
How to Fix a Car Horn YourMechanic Advice
How to Wire a Car Horn It Still Runs

HornBlasters Don’t Blow Your Temper Blow Your Horns!
How to wire in an extra car horn with a switch on the dash
How to Wire and Install Hella Horns 1A Auto
How to Install Aftermarket Hella Horns YouTube
Our 1A Auto Video Library has thousands of how-to auto repair installation videos specific to year, make and model. Each video guides you with step-by-step instructions to empower you to tackle your own repairs on your car, truck, SUV, or van.
Welcome to HELLA Horns. Please choose your region and language! Europe deutsch english français italiano español nederlands türkçe. Asia english 中国的
Mar 08, 2016 · You will need to show us the photo of the stock horn wiring or a complete wiring diagram of the car to advise you whether the stock horn comes with relay. You want to buy a pen, you must also let us know the specs. Like type of ink, colour required, rubber grip, cap type or click type.
Oct 18, 2018 · Buy Wolo (430) Musical Horn – 12 Volt, Plays Dixieland: Air Horns – Amazon.com FREE Kindly refer the “Installation Instructions” and the “Product Description” given below for troubleshooting steps. minus the horns, and run the compressor, once with the car off, and once with the car fully on. If it works with the car off, it doesn’t
Mar 18, 2018 · Buy GAMPRO 12V 150db Air Horn, Chrome Zinc Dual Trumpet Air Horn with Compressor for Any 12V Vehicles Trucks Lorrys Trains Boats Cars Vans: Air Horns – Amazon.com FREE DELIVERY possible on eligible purchases
Replace your old horn in your car, boat, RV or other vehicle with this set of 12 volt electric horns, fashioned in the classic snail shape. Constructed of high-impact thermoplastic on the inside and chrome on the outside, the electric horns generate a blast that exceeds 110 decibels. Mounting tabs, brackets and installation instructions included.
Mar 30, 2016 · How to Fix a Car Horn by Mia Bevacqua on March 30, 2016 Schedule Horn does not blow Inspection. Install the new fuse – If you find the fuse has failed, install a new one of the same amperage rating. To do this, simply push the fuse back into its correct slot. Test the ground side of the circuit – According to your car’s manual
HWK-1 comes with wire harness, quick relay connect plug, terminals, in-line fuse, plastic wire ties and easy to follow instructions in English and Spanish. Wolo’s new wiring kit makes installation of any low pressure air horn easy
INSTALLING AFTERMARKET HORNS – ET-208. Check this diagram first to remind yourself of the original horn wiring. You will notice that power is supplied to the horn from the fuse which is always hot (not switched), so the horn will work even when the key is switched off.
Our horns can also be hooked up to a 12 volt lantern battery for portability. Remote control comes complete with installed battery and operates up to a range of 10-20 feet. Quick and easy installation…takes about 5 minutes to install…instructions are included. This is the gift of a lifetime!
May 30, 2013 · How To Wire Air Horns / Air Horn Install for automotive and marine use *Includes Install instructions. Air Horn Car Install and Relay Wiring Instructions – Duration:
Train Horns, Air Horns, Onboard Air Systems, and Air Suspension, we have it all! We bring the real sound of a locomotive right to your vehicle and service all your pneumatic needs. Our Horn Kits are simply ridiculous and provide your vehicle with a voice on the road!
The Stebel Nautilus is one of my personal favorite car horns and I have owned several over the years. It has a unique sound from the twin tone harmonized horn that outputs at 139dB.It is a compressor based car horn (water resistant) that has a compact design to fit in all locations under the bonnet.
Viking Horns
How to Install Aftermarket Hella Horns YouTube
GMTRK-2 BOLT-ON TRAIN HORN SYSTEM INSTALLATION INSTRUCTIONS For 2007-2013 GM & GMC Trucks & SUVS The GMTRK-2 System comes pre-assembled with air compressor, air tank and all fittings installed in the tank and on the battery tray plate. The horns will be bolted to the tow hook mount, making installation fast & efficient.
Hella® – Horns Installation Instructions Warranty Hella, Inc.’s limited warranty guarantees Hella products to be free from defects in materials and workmanship for the period of (1) one year after retail purchase from an authorized Hella dealer.
Find a good spot under the hood to install the horn(s). The ideal mounting position is towards the front of your vehicle, and pointing a bit downward so that water doesn’t collect in the horn. Mount the horn to your vehicle, using the supplied mounting hardware and screws. If necessary, install the included compressor to your firewall or a fender. Hook the compressor up to the horn with the included tubing, making sure …
Replace your old horn in your car, boat, RV or other vehicle with this set of 12 volt electric horns, fashioned in the classic snail shape. Constructed of high-impact thermoplastic on the inside and chrome on the outside, the electric horns generate a blast that exceeds 110 decibels. Mounting tabs, brackets and installation instructions included.
HornBlasters Instruction Manuals and Schematics. Lose your manual or want a digital copy? We got your back! Most manuals are provided in Adobe PDF format only.
Car horns and trumpet horns The HELLA Group specializes in innovative lighting systems and vehicle electronics. It has been one of the technology leaders over the last hundred years and an important partner of the automotive industry, the independent aftermarket and for workshops.
Jun 26, 2017 · Air horn setups usually contain multiple horn pieces that emit sound. Because these pieces are trumpet-shaped, we’ll refer to them simply as “trumpets”. For example, kits we offer contain between 1 and 5 trumpets. Most of them also include a small air compressor. This detail will be noted on the product page.
Mar 08, 2016 · You will need to show us the photo of the stock horn wiring or a complete wiring diagram of the car to advise you whether the stock horn comes with relay. You want to buy a pen, you must also let us know the specs. Like type of ink, colour required, rubber grip, cap type or click type.
Mar 18, 2018 · Buy GAMPRO 12V 150db Air Horn, Chrome Zinc Dual Trumpet Air Horn with Compressor for Any 12V Vehicles Trucks Lorrys Trains Boats Cars Vans: Air Horns – Amazon.com FREE DELIVERY possible on eligible purchases
Components and Installation Instructions General Information: Your vehicle must have a 12 volt power source to hook up this horn unit. (NOTE: Our horns are NOT waterproof, so be sure to mount it in a dry location) You can wrap your horn in a plastic bag after it is …
May 30, 2013 · How To Wire Air Horns / Air Horn Install for automotive and marine use *Includes Install instructions. Air Horn Car Install and Relay Wiring Instructions – Duration:
Mar 08, 2015 · No need to disconnect the battery. If your car horn max at 11A, while your horn fuse is at 10A, you can just swop out the fuse for 15A one. I did that for my Stebel Magnum horns, been using them for the past 6 years without problem. If you are worried the wires may burn, then you can get the workshop to install the relay.
Dec 05, 2018 · How to Install an Air Horn Step 1. Open the hood of your vehicle and choose a good place to mount the horns. Step 2. Using self-tapping screws and the provided mounting hardware,… Step 3. Mount the compressor to a fender or firewall with the appropriate hardware,… Step 4. Use a …
Dec 18, 2017 · Air Horn Car Install and Relay Wiring Instructions – Duration: 6:26. Strange Garage 83,143 views
Our horns can also be hooked up to a 12 volt lantern battery for portability. Remote control comes complete with installed battery and operates up to a range of 10-20 feet. Quick and easy installation…takes about 5 minutes to install…instructions are included. This is the gift of a lifetime!
Godzilla Roar Sounds Car Horn Wireless Boom Blasters
Viking Horns
Oct 09, 2019 · How to Install a Car Alarm. Car alarms are an effective way to insure your investment and give you peace of mind. While many cars come standard with some kind of alarm system, some do not. It is no surprise that many people want to install…
Car horn installation using the example of an Audi TT/RS: Instructions Preparatory work The power supply of the acoustic signaling device is to be cut off throughout the entire setting up of the electrical connection.
How to Install the Air Horn on a Silverado Truck by Mike Frees . If you are not satisfied with the sound of the horn in your Chevy Silverado, replacing it with an air horn is not a difficult task. A variety of horns are available; some are one piece, with the horn and compressor assembled together. Wolo Manufacturing: Installation
Dec 05, 2018 · How to Install an Air Horn Step 1. Open the hood of your vehicle and choose a good place to mount the horns. Step 2. Using self-tapping screws and the provided mounting hardware,… Step 3. Mount the compressor to a fender or firewall with the appropriate hardware,… Step 4. Use a …
Oct 18, 2018 · Buy Wolo (430) Musical Horn – 12 Volt, Plays Dixieland: Air Horns – Amazon.com FREE Kindly refer the “Installation Instructions” and the “Product Description” given below for troubleshooting steps. minus the horns, and run the compressor, once with the car off, and once with the car fully on. If it works with the car off, it doesn’t
May 30, 2013 · How To Wire Air Horns / Air Horn Install for automotive and marine use *Includes Install instructions. Air Horn Car Install and Relay Wiring Instructions – Duration:
Aug 10, 2016 · In this video, the little broski and I install some new horns on the Golf. The stock horns on the car worked, however they weren’t loud enough for our liking, so we decided to pick up a set of
Mar 30, 2016 · How to Fix a Car Horn by Mia Bevacqua on March 30, 2016 Schedule Horn does not blow Inspection. Install the new fuse – If you find the fuse has failed, install a new one of the same amperage rating. To do this, simply push the fuse back into its correct slot. Test the ground side of the circuit – According to your car’s manual
Replace your old horn in your car, boat, RV or other vehicle with this set of 12 volt electric horns, fashioned in the classic snail shape. Constructed of high-impact thermoplastic on the inside and chrome on the outside, the electric horns generate a blast that exceeds 110 decibels. Mounting tabs, brackets and installation instructions included.
Car horns and trumpet horns The HELLA Group specializes in innovative lighting systems and vehicle electronics. It has been one of the technology leaders over the last hundred years and an important partner of the automotive industry, the independent aftermarket and for workshops.
Using the self-tapping screw, mount the 20-amp circuit breaker in a protected location near the vehicle battery. Step 3 Connect one terminal on the circuit breaker to the positive terminal of the battery, using a piece of wire and two solderless ring terminals. Step 4
Welcome to HELLA Horns. Please choose your region and language! Europe deutsch english français italiano español nederlands türkçe. Asia english 中国的
Godzilla Roar Sounds Wireless Car Horn. These horns have a small design…but packs a big powerful sound. 20 watts output sound — 119 decibels…these horns are LOUD! Quick and easy installation…takes about 5 minutes to install…instructions are included.
Dec 18, 2017 · Air Horn Car Install and Relay Wiring Instructions – Duration: 6:26. Strange Garage 83,143 views
How to Wire a Car Horn It Still Runs
Car horn installation General Car Discussion
Mar 18, 2018 · Buy GAMPRO 12V 150db Air Horn, Chrome Zinc Dual Trumpet Air Horn with Compressor for Any 12V Vehicles Trucks Lorrys Trains Boats Cars Vans: Air Horns – Amazon.com FREE DELIVERY possible on eligible purchases
Feb 21, 2015 · basic wiring guys, hope you guys like whats going on, we will make progress together!! i will show a more advanced circuit with relays in the future.
Mar 08, 2016 · You will need to show us the photo of the stock horn wiring or a complete wiring diagram of the car to advise you whether the stock horn comes with relay. You want to buy a pen, you must also let us know the specs. Like type of ink, colour required, rubber grip, cap type or click type.
Mar 08, 2015 · No need to disconnect the battery. If your car horn max at 11A, while your horn fuse is at 10A, you can just swop out the fuse for 15A one. I did that for my Stebel Magnum horns, been using them for the past 6 years without problem. If you are worried the wires may burn, then you can get the workshop to install the relay.
Replace your old horn in your car, boat, RV or other vehicle with this set of 12 volt electric horns, fashioned in the classic snail shape. Constructed of high-impact thermoplastic on the inside and chrome on the outside, the electric horns generate a blast that exceeds 110 decibels. Mounting tabs, brackets and installation instructions included.
Find a good spot under the hood to install the horn(s). The ideal mounting position is towards the front of your vehicle, and pointing a bit downward so that water doesn’t collect in the horn. Mount the horn to your vehicle, using the supplied mounting hardware and screws. If necessary, install the included compressor to your firewall or a fender. Hook the compressor up to the horn with the included tubing, making sure …
Welcome to HELLA Horns. Please choose your region and language! Europe deutsch english français italiano español nederlands türkçe. Asia english 中国的
Instructions for Club Car DS Models Figure 2 Figure 1 (28905)! 2 Use part number 6439 turn signal head, that will have a corresponding nine pin connector and horn button for a plug and play installation. Use the supplied brake lead harness for switch connection. On some cars you may
Car horn installation using the example of an Audi TT/RS: Instructions Preparatory work The power supply of the acoustic signaling device is to be cut off throughout the entire setting up of the electrical connection.
Jun 26, 2017 · Air horn setups usually contain multiple horn pieces that emit sound. Because these pieces are trumpet-shaped, we’ll refer to them simply as “trumpets”. For example, kits we offer contain between 1 and 5 trumpets. Most of them also include a small air compressor. This detail will be noted on the product page.